Diagnosis, classification, symptoms, and causes of hypersomnias

What are hypersomnias?
According to the ICSD-3-TR, hypersomnias are a category of chronic neurologic sleep disorders that are also called central disorders of hypersomnolence (CDH). They cause people to sleep excessive amounts (long sleep), have excessive daytime sleepiness (EDS), or both. EDS is a strong daytime sleepiness or need to sleep during the day, even with enough sleep the night before. These disorders are often debilitating, significantly affecting social, school, and occupational functioning. Symptoms and their severity may fluctuate.
Refer your patients to our web page “Read about hypersomnia sleep disorders.”
How are hypersomnias classified?
Hypersomnias may be classified as primary or secondary. Primary means it occurs on its own and isn’t caused by another condition. Secondary means it’s caused by or closely related to a different condition.
| Primary hypersomnias |
|---|
| Idiopathic hypersomnia (IH) with and without long sleep |
| Narcolepsy Type (NT2) without cataplexy |
| Narcolepsy Type 1 (NT1) with cataplexy |
| Kleine-Levin syndrome (KLS) |
| Secondary hypersomnias |
|---|
| Hypersomnia associated with a medical disorder, such as a head injury, a neurodegenerative disease such as Parkinson’s disease, or a neuromuscular disorder such as myotonic dystrophy |
| Hypersomnia associated with a medicine or substance – although the ICSD-3-TR still labels this as hypersomnia “due to” a medicine or substance, the ICSD committee intended to change to “associated with” because it’s often impossible to be sure that a certain medicine or substance is the cause or is the only cause |
| Insufficient sleep syndrome – sleepiness due to regularly not sleeping enough hours each night (7 to 9 hours for adults) |
| Hypersomnia associated with a mental disorder, such as depression or seasonal affective disorder |
What are the symptoms and diagnostic criteria of the primary hypersomnias?
Symptoms
The average age of symptom onset is 16 to 21 years old. In addition to EDS, symptoms of idiopathic hypersomnia (IH) may include:
- Long unrefreshing naps — these may be hard or impossible to avoid, are usually more than 1 hour, and may make people feel even worse
- Brain fog
- Long sleep — needing at least 11 hours of sleep per 24-hour period (including naps) or more than 9 hours at night (or during the main sleep period)
- Sleep drunkenness (or severe sleep inertia)
- Unrefreshing sleep
- Sleep attacks
- Fatigue
- Automatic behaviors
- Autonomic symptoms, such as headache, orthostatic intolerance, and temperature dysregulation
- Motor hyperactivity
- Sleep-related hallucinations
- Sleep paralysis
- Disrupted nighttime sleep
Summarized ICSD-3-TR diagnostic criteria for idiopathic hypersomnia (IH)
The ICSD-3-TR diagnostic criteria don’t differentiate between idiopathic hypersomnia (IH) with and without long sleep. The criteria are partly a diagnosis of exclusion because researchers don’t know the cause of IH or its biomarkers. The criteria are:
- Daily EDS for at least 3 months
- Overnight PSG and MSLT results don’t indicate Narcolepsy Type 1 or Narcolepsy Type 2
- No more than 1 SOREMP on the MSLT and preceding overnight PSG combined
- No cataplexy
- The presence of one or both of the following:
- MSLT shows a mean sleep latency of 8 minutes or less
- Total 24-hour sleep time is at least 11 hours (more typically 12 to 14 hours) when measured in one of the following ways:
- 24-hour PSG after correcting any chronic sleep deprivation
- Wrist actigraphy recorded along with a sleep log and averaged over at least 7 days of unrestricted sleep
- Sufficient sleep for at least 1 week (preferably confirmed by wrist actigraphy) fails to improve sleepiness
- In people with sleep needs of more than 7 to 8 hours, average sleep duration (for example, 7 hours per night in adults) may be insufficient
- Long sleepers don’t have EDS if they are allowed to sleep as long as they need
- Another sleep disorder, medical or mental disorder, or medicine or substance use doesn’t better explain the symptoms
Note: The total 24-hour sleep time of 11 hours may need to be modified to account for cultural variability and stages of child development. For example, the cutoff needs to be longer in school-age children (age 6 to 13) who normally sleep 9 to 12 hours in 24. For an individual child, it’s also important to take into consideration changes from their historical normal.
Additional supporting features:
- Sleep drunkenness
- Naps that are unrefreshing and long (more than 1 hour)
Sometimes people who have significant symptom severity may not meet the full ICSD-3-TR diagnostic criteria for idiopathic hypersomnia (IH). If so, use clinical judgment to diagnose idiopathic hypersomnia (IH). Take great care to continue to rule out other conditions, and consider repeating sleep studies.
Summarized DSM-5-TR diagnostic criteria
The closest match to idiopathic hypersomnia (IH) in the DSM-5-TR is hypersomnolence disorder. The diagnosis of hypersomnolence disorder requires the following:
- EDS or hypersomnolence despite a main sleep period lasting at least 7 hours and also associated with one or more of the following:
- Additional recurrent sleep periods or lapses during the day
- A main sleep period that is greater than 9 hours and unrefreshing
- Abrupt awakening leads to difficulty becoming fully awake
- Significant impairment in occupational, cognitive, social, or other important functioning
- EDS happens at least 3 times a week and lasts for at least 3 months
- Hypersomnolence isn’t better explained by another sleep disorder
- When other mental or medical conditions coexist, they don’t fully explain the hypersomnolence
- Hypersomnolence isn’t caused by a medicine or substance abuse
Download a printable fact sheet with IH characteristics and diagnostic criteria.
Symptoms
Symptoms usually start during adolescence. In addition to EDS, symptoms of Narcolepsy Type 2 may include:
- Brain fog
- Disrupted nighttime sleep
- Required naps — these may be hard or impossible to avoid and are usually short and somewhat refreshing
- Sleep-related hallucinations
- Sleep paralysis
- Sleep attacks
- Fatigue
- Sleep drunkenness (or severe sleep inertia)
- Long sleep — needing at least 11 hours of sleep per 24-hour period (including naps) or more than 9 hours at night (or during the main sleep period)
- Automatic behaviors
- Autonomic symptoms, such as headache, orthostatic intolerance, and temperature dysregulation
- Obesity
- Motor hyperactivity
Summarized ICSD-3-TR diagnostic criteria for Narcolepsy Type 2
The ICSD-3-TR criteria are partly a diagnosis of exclusion because researchers don’t know the cause of Narcolepsy Type 2 or its biomarkers. The criteria are:
- Daily EDS for at least 3 months
- MSLT shows a mean sleep latency of 8 minutes or less (typically less than 5 minutes) and 2 or more SOREMPs
- No cataplexy (if it develops later, change the diagnosis to Narcolepsy Type 1)
- If you test CSF orexin it’s either more than 110 pg/mL (when using a Stanford reference sample) or more than one-third of mean values from normal subjects using the same test
- Symptoms aren’t better explained by another sleep disorder, medical or mental disorder, medicine or substance use, and particularly not by insufficient sleep.
Symptoms
The usual age of symptom onset is 5 to 25 years old. In addition to EDS, symptoms of Narcolepsy Type 1 may include:
- Cataplexy
- Required naps — These may be hard or impossible to avoid but are usually short and refreshing
- Disrupted nighttime sleep
- Sleep-related hallucinations
- Sleep paralysis
- Brain fog
- Sleep attacks
- Obesity
- Fatigue
- Automatic behaviors
- Autonomic symptoms, such as headache, orthostatic intolerance, and temperature dysregulation
- Frequent vivid dreams or nightmares
- Motor hyperactivity
Summarized ICSD-3-TR diagnostic criteria for Narcolepsy Type 1
Narcolepsy Type 1 is caused by problems with central nervous system orexin signaling, which leads to REM sleep instability, including cataplexy. Therefore, the following criteria must be met:
- Daily EDS
- One or more of the following:
- Typical cataplexy and either:
- MSLT shows a mean sleep latency of 8 minutes or less (typically less than 5 minutes) and 2 or more SOREMPs
- A SOREMP (within 15 minutes of sleep onset) on overnight PSG
- CSF orexin is 110 pg/mL or less (using a Stanford reference sample) or less than one-third of mean values from normal subjects using the same test
- Typical cataplexy and either:
- Symptoms aren’t better explained by another sleep disorder, insufficient sleep, medical or mental disorder, or medicine or substance use
Typical cataplexy is often critical for NT1 diagnosis because about 95% of people with it have low CSF orexin. It’s very important to differentiate typical cataplexy from atypical cataplexy and other mimics, as detailed below.
Watch this short BBC video of a cataplexy episode:
What are the features of typical cataplexy?
- Retained consciousness
- Sudden loss of muscle tone with sudden return of muscle tone after the episode
- Loss of muscle tone is bilaterally symmetrical
- Reversible loss of deep tendon reflexes in involved limbs during episodes
- Respiratory muscles aren’t involved, but people sometimes have shortness of breath
- Triggered by strong emotions, usually positive, such as the emotions associated with laughter
- Brief — lasts less than 2 minutes (usually less than 30 seconds)
- Variable frequency — at least 3 lifetime episodes ranging from once every few months to more than 20 per day (usually at least 1 episode a month if untreated)
- Improvement with medicines that treat cataplexy
- Can be partial or complete cataplexy, but usually partial
What’s the difference between partial and complete cataplexy?
Partial cataplexy
Episodes happen in specific parts of the body. It can be very subtle, sometimes lasting only a few seconds and only recognized by experienced observers such as the patient’s partner. When episodes are longer, there is bilateral loss of muscle tone in the face, neck, or legs, and sometimes also the arms. Examples include:
- Buckling knees
- Head drop
- Facial weakness that may lead to a sagging jaw and difficulty speaking
Complete cataplexy
This is less common. Many parts of the body or sometimes the entire body are affected. It builds up over several seconds, usually starting as a partial episode in the face or neck, and may progress to complete weakness and collapse. Sometimes people may fall asleep during prolonged episodes.
When does cataplexy last more than 2 minutes?
Cataplexy usually lasts less than 2 minutes. It can last longer in these situations:
- If a particular trigger continues, attacks can merge and appear to be one long episode
- If a person with NT1 suddenly stops taking medicine that treats cataplexy, especially antidepressants, this can cause “status cataplecticus,” in which long-lasting episodes happen almost continuously
What are some variations of typical cataplexy?
People who have typical cataplexy may sometimes also have these variations:
- Spontaneous episodes that aren’t clearly triggered by emotions — these usually happen in the evening, and sleepiness makes them more likely
- Muscle weakness may be asymmetrical (stronger on one side than the other), but not unilateral (only on one side of the body)
- Facial twitching
- Jerky arm movements
What is atypical cataplexy?
Atypical cataplexy is much less strongly associated with orexin deficiency and NT1 diagnosis. Episodes shouldn’t be considered typical cataplexy if 2 or more of these features are present:
- Uncertainty about retained consciousness
- Very fast generalized muscle weakness without build-up over seconds, leading to falls and injuries
- Loss of muscle tone is only unilateral
- Episodes are never triggered by funny situations or laughter
- Absence of clear triggers, or the only triggers are negative emotions (such as anxiety, fear, or a sudden, startling noise)
- Episodes in adults last longer than 3 minutes without an ongoing trigger
- Prolonged recovery time (several minutes) needed after an episode
- Episodes of complete cataplexy only (without a history of partial cataplexy)
What else might mimic cataplexy?
- Syncope
- Transient ischemic attacks
- Drop attacks
- Akinetic seizures
- Neuromuscular disorders
- Vestibular disorders
- Psychological and psychiatric disorders
- Dozing (such as sleep attacks) and sleep paralysis
Don’t diagnose cataplexy if any of these features are present:
- Loss of consciousness from the start of the episode
- Episodes last longer than 10 minutes without a trigger
- Normal deep tendon reflexes of the involved limb during an observed episode
- Clear signs of episodes other than cataplexy (such as typical aura of epilepsy or typical prodromal signs before vasovagal syncope)
- Symptoms may be more difficult to assess, especially depending on the child’s verbal ability.
- The MSLT may be more difficult to perform because of the unfamiliar sleep environment and/or difficulty remaining awake between MSLT nap opportunities. Naps are normal for young children under age 5 to 6 years and can make study interpretation difficult.
- Sudden onset in days to weeks is more common in children than in adults.
- Sleepiness may appear as excessively long night sleep or the recurrence of daytime napping after the child stopped needing naps.
- Sleepiness can present as behavioral problems, including hyperactivity, inattentiveness, fidgeting, impulsivity, and emotional lability.
What are some common misdiagnoses in children?
Because sleepiness can present as inattention, impulsiveness, and/or hyperactivity, NT1 in children may be misdiagnosed as ADHD. Other conditions that NT1 can be misdiagnosed as include seizure disorder, schizophrenia, depression, autism spectrum disorder, insomnia, or obstructive sleep apnea. This can happen because of misinterpreting narcolepsy symptoms such as cataplexy, sleep-related hallucinations, social withdrawal due to sleepiness, or disrupted nighttime sleep. This is further complicated by the fact that these other conditions may be comorbid with narcolepsy.
How can cataplexy be different in children?
Cataplexy in children:
- Can be very severe near disease onset
- Is usually triggered by emotions that may only happen in certain social settings or environments (such as hearty laughter induced by a certain friend or friend group)
- Can present as static cataplexy
- Cataplexy can be constant and not just triggered by emotion
- Cataplectic facies has been described, with weakness in the face, eyelids, and mouth, including tongue protrusion
- Cataplexy can present with an unsteady hypotonic ataxia-like gait
- Can present as positive motor phenomena (spontaneous muscle hyperactivity), especially facial muscle twitching or dyskinesias (uncontrollable body movements that can affect just one part of the body, like the face or arms, or the entire body)
Additional symptoms for children may include:
- Early puberty
- Rapid-onset obesity close to disease onset
Additional resources
- Clinical Symptoms of Children vs. Adults With Narcolepsy and Cataplexy: An overview of the main differences in NT1 symptoms between those under and over 18 years old, including a link to the source journal article
- Recognize Narcolepsy in Pediatric Patients: Detailed descriptions of symptom presentation, including cataplexy videos and screening and clinical interview tools
Symptoms
Symptoms usually start in early adolescence. Kleine-Levin syndrome is defined by repeated episodes of severe excessive daytime sleepiness and sleep duration, with cognitive, psychiatric, and behavioral symptoms:
- Very long sleep
- Sleeping much longer than usual, up to 16 to 20 hours per day
- Waking or getting up only to eat and go to the bathroom
- Arousable but irritable if kept from sleeping
- Extreme fatigue is to the point of being bedridden with no action
- A typical episode starts suddenly and lasts a median of 10 days (range, 2.5 to 80 days)
- Episodes last several weeks to months in 30% of people with KLS
- During episodes, normal daily activities stop and people are unable to care for themselves
- Triggers include flu-like illness, infection of the upper airway, gastroenteritis, alcohol consumption, sleep deprivation, stress, travel, head trauma, and menstruation
| In addition to excessive daytime sleepiness and sleep duration, symptoms of Kleine-Levin syndrome during episodes include (at least one): |
|---|
| Anterograde amnesia |
| Major apathy |
| Confusion in time (and sometimes in space) |
| Slow speech |
| Derealization |
| Hyperphagia |
| Hypersexuality (usually in males) |
| Other possible symptoms include: |
|---|
| Childlike demeanor |
| Hallucinations and delusions |
| Headaches |
| Hypersomnia associated with a mental disorder, such as depression or seasonal affective disorder |
Summarized ICSD-3-TR diagnostic criteria for Kleine-Levin syndrome
The following criteria must be met:
- At least 2 episodes of excessive sleepiness and sleep duration, each lasting for 2 days to several weeks
- Episodes usually happen more than once a year and at least once every 18 months (median 3 months) for many years
- Between episodes, at least during the first few years of illness, sleep and wakefulness, cognition, behavior, and mood are usually normal or near normal
- However, sometimes between episodes, sleep duration may be longer, cognition may be mildly altered, and some mental disorders may be evident
- At least one of the following happens during episodes:
- Cognitive dysfunction
- Major apathy
- Derealization
- Disinhibited behavior (such as hypersexuality or hyperphagia)
- Symptoms aren’t better explained by another sleep disorder, insufficient sleep, medical or mental disorder, or medicine or substance use
What might mimic Kleine-Levin syndrome?
- Structural neurological disorders, such as tumors within the 3rd ventricle may obstruct ventricular flow
- This can lead to headaches, vomiting, sensory disturbances, and episodes of impaired alertness
- Encephalitis
- Hyperammonemic encephalopathy
- Multiple sclerosis
- Head trauma
- Porphyria
- Lyme disease
- Basilar migraine
- Complex partial status epilepticus
- Certain mental disorders:
- Recurrent episodes of sleepiness are also reported as a symptom of mental disorders such as depression, bipolar disorder, seasonal affective disorder, and somatoform disorder
- However, the onset and offset of symptoms are typically less abrupt than in KLS, and presenting symptoms may differ
Who is more likely to have a longer Kleine-Levin syndrome duration?
Several long-term studies show an often-improving course, with episodes lessening in frequency, duration, and severity over the course of about 14 years. However, longer disease duration is predicted by:
- Male sex
- Age at onset younger than 12 years or older than 20 years
- Hypersexuality during episodes
When should I diagnose menstrual-related hypersomnia?
Also known as menstrual-related Kleine-Levin syndrome, use this diagnosis when hypersomnolence episodes only happen just before or during menses. These episodes:
- Last 3 to 15 days
- Happen less than 3 times a year
- Have been associated with compulsive eating, sexual disinhibition, and depressive mood
Treatment with oral birth control pills may help.
For correct interpretation of overnight PSGs and MSLTs, recordings need to meet the following standard conditions:
- Before in-lab sleep tests, check for insufficient sleep and circadian rhythm disturbances using a sleep diary (with actigraphy when possible)
- Download the American Academy of Sleep Medicine’s (AASM’s) Two Week Sleep Diary
- Standardize the sleep-wake schedule and ensure a minimum of 7 hours in bed each night (ideally more), starting at least 7 days before testing
- Because the circadian clock strongly affects REM sleep, shift workers need to stabilize on a normal schedule before you can accurately diagnose them with NT2
- Allow for appropriate sleep duration for children and long sleepers, which may be longer
- Stop medicines that influence sleep propensity and architecture at least 14 days (or at least 5 times the half-life of the medicine and longer-acting metabolite) before testing
- Some medicines, like antidepressants, may need to be slowly tapered and slowly restarted and therefore will be ineffective for a longer time
- In certain situations, such as a history of illegal drug use, you may want to check a urine drug screen
- Do the overnight PSG on the night immediately before the MSLT
Follow AASM guidelines:
- “Manual for the Scoring of Sleep and Associated Events”
- “Recommended protocols for the MSLT and MWT in adults”
Who might need medical leave for their sleep studies?
To meet these test protocols, you may need to help people access medical leave, such as FMLA or short-term disability, for a couple weeks from their work or school. This is especially the case for:
- People who need to stop one or more medicines before testing
- Children and long sleepers who need to get more than 7 hours of sleep at night for 1 to 2 weeks before testing
- Shift workers
Refer your patients to our web pages:
- Going to school while coping with a hypersomnia
- Planning for job accommodations and disability income
When should I repeat sleep studies?
- Once you diagnose NT1, you usually don’t need to repeat sleep studies.
- For people with other hypersomnias, consider repeating sleep studies only if absolutely necessary — for example, if symptoms have changed significantly and you suspect the diagnosis has also changed.
- When insurers ask for repeat sleep studies, let them know if you don’t think these are medically indicated, or consider asking if you can substitute actigraphy with a detailed sleep diary or other testing that’s less difficult for your patients.
Consider ordering HLA-DQB1 blood typing before doing a spinal tap to measure CSF orexin. If HLA-DQB1*06:02 is negative, CSF orexin will almost certainly be normal. In very rare cases, other rare HLA types are positive, and low orexin levels can happen (Miano, 2023).
Consider testing CSF orexin levels if the:
- Cataplexy history isn’t clear
- Person is of African descent, since narcolepsy without cataplexy but with orexin deficiency may be more frequent
- MSLT interpretation is complicated by sleep apnea, a mental disorder, or medicines that influence sleep
You should diagnose NT1 if the CSF orexin level is 110 pg/mL or less (using a Stanford reference sample) or less than one-third of mean values from normal subjects using the same test.
In the U.S., you can get these tests by sending your samples to Mayo Clinic Labs:
For more information, visit Sleep Review Magazine’s article “Mayo Clinic Can Test Your Patients’ Orexin Levels.”
Idiopathic hypersomnia (IH) with and without long sleep time may be separate diagnoses
Recent studies show that idiopathic hypersomnia (IH) with and without long sleep time should be considered as separate diagnoses:
- An analysis that clustered people with hypersomnias into groups based on common symptoms found idiopathic hypersomnia (IH) with long sleep time formed a clear group. The same analysis found idiopathic hypersomnia (IH) without long sleep time in a different group (Sonka, 2015).
- Another cluster analysis also found a clear group with a more severe form of Iidiopathic hypersomnia (IH) associated with longer sleep duration and worse EDS, sleep inertia, functional impairment, and depressive symptoms (Cook, 2019).
- Another analysis found 2 distinct clusters of people without cataplexy who significantly differed in presence of sleep drunkenness, subjective difficulty awakening, and weekend-week sleep length difference (Gool, 2022).
Idiopathic hypersomnia (IH) without long sleep and Narcolepsy Type 2 can overlap
Cluster analyses sort into the same group:
- People with Narcolepsy Type 2
- People with idiopathic hypersomnia (IH) without long sleep time (Sonka, 2015; Gool, 2022)
The only diagnostic criteria that distinguishes between idiopathic hypersomnia (IH) and Narcolepsy Type 2 is that REM sleep in MSLT nap tests happens in Narcolepsy Type 2 but not in idiopathic hypersomnia (IH).
However, multiple studies have shown that if you repeat the MSLT:
- Results can change, especially for people with disorders other than Narcolepsy Type 1
- The same person might be diagnosed with idiopathic hypersomnia (IH), Narcolepsy Type 2, or sometimes not meet diagnostic criteria for either
Narcolepsy Type 1 and Narcolepsy Type 2 can be difficult to differentiate from each other
People who are initially diagnosed with Narcolepsy Type 2 may eventually show cataplexy or low orexin levels. If this happens, change their diagnosis to Narcolepsy Type 1.
Symptoms of idiopathic hypersomnia (IH) with and without long sleep, Narcolepsy Type 1, and Narcolepsy Type 2 overlap
Each of these disorders causes EDS and brain fog. The table below shows how common the other symptoms are, depending on the type of sleep disorder. (Darker blue means a higher percentage of people have these symptoms.)
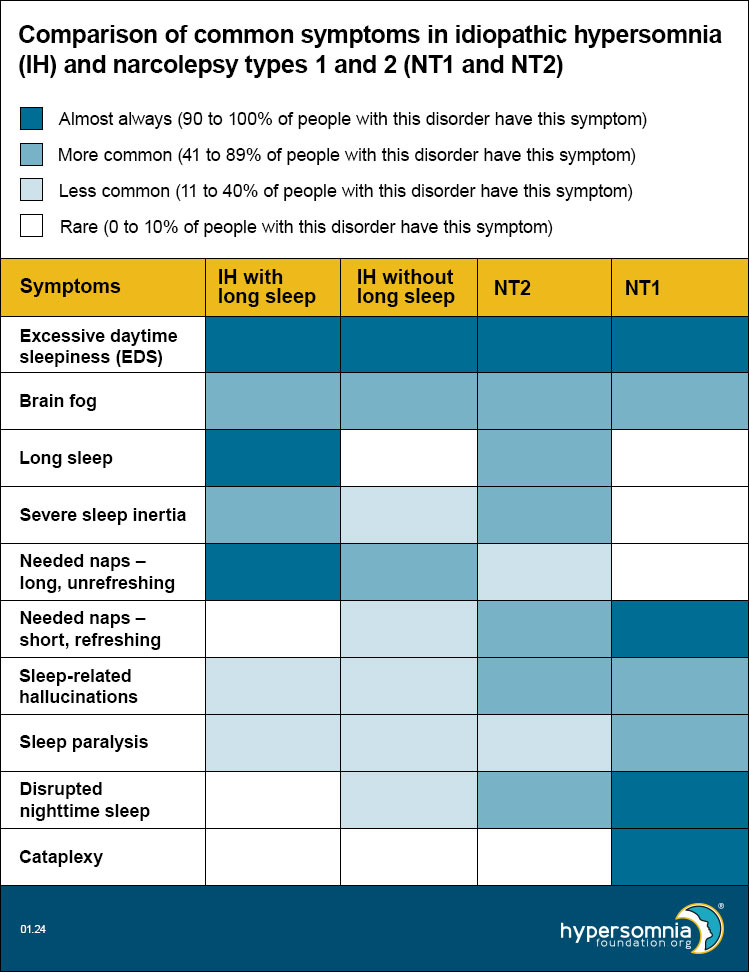
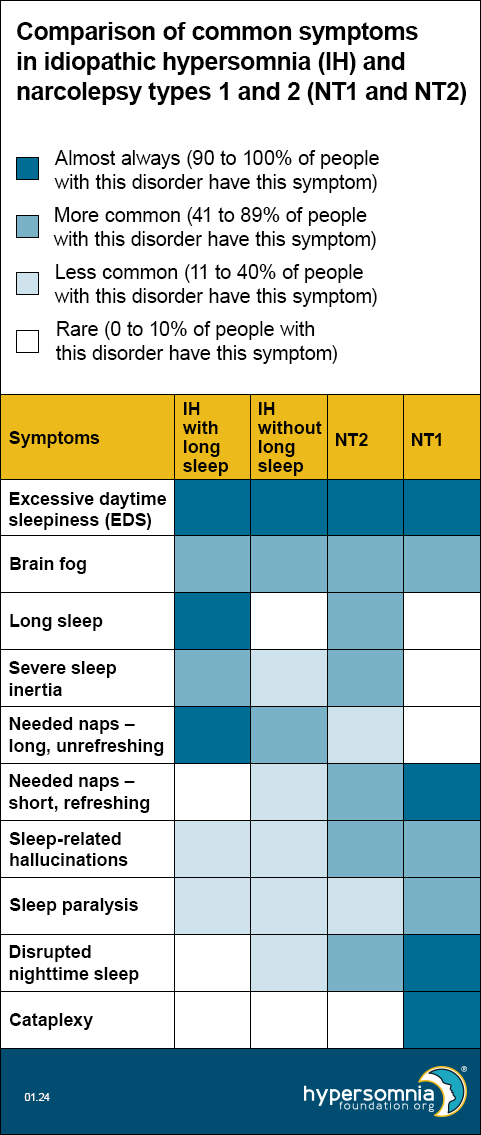
Table notes and references
- EDS — Present by definition, so 100% for all.
- Brain fog — Per unpublished 2023 HF CoRDS data, 53% of people with idiopathic hypersomnia (IH) have brain fog. Per Maski 2017, 47% of people with Narcolepsy Type 1 and 53% of people with Narcolepsy Type 2 had cognitive difficulties.
- Long sleep — For idiopathic hypersomnia (IH) with and without long sleep, these are 100% and 0% respectively by definition. Per Vernet 2009 and Broughton 1988, long sleep is less than 10% for Narcolepsy Type1. Per Barateau 2024, long sleep is 76% for Narcolepsy Type2.
- Severe sleep inertia — Per ICSD-3-TR, 36-66% of idiopathic hypersomnia (IH) (doesn’t differentiate long sleep); patients with long sleep duration have higher rates of sleep drunkenness and unrefreshing naps, so we used the top end of the range for idiopathic hypersomnia (IH) with long sleep and the bottom end of the range for idiopathic hypersomnia (IH) without long sleep. Per Khan 2015, severe sleep inertia is rare, but occasionally reported in Narcolepsy Type 1, may be common in Narcolepsy Type 2, and is common in idiopathic hypersomnia (IH). Per Barateau 2024, sleep inertia is 83% in NT2.
- Long unrefreshing naps — Per ICSD-3-TR, 46-78% of people with idiopathic hypersomnia (IH); patients with long sleep duration have higher rates of sleep drunkenness and unrefreshing naps, so we used the top end of the range for idiopathic hypersomnia (IH) with long sleep and the bottom end of the range for idiopathic hypersomnia (IH) without long sleep.
- Refreshing (restorative) naps — Per ICSD-3-TR, refreshing daytime naps are characteristic of Narcolepsy Type 2. Per Khan 2015, they’re common in Narcolepsy Type 1, but naps in idiopathic hypersomnia (IH) are long and unrefreshing.
- Sleep-related hallucinations — Per ICSD-3-TR, 4-40% in idiopathic hypersomnia (IH) and 33-80% in narcolepsy. Per Khan 2015, 77% of Narcolepsy Type 1, 42% of Narcolepsy Type 2, and 25% of idiopathic hypersomnia (IH).
- Sleep paralysis — Per ICSD-3-TR: 4-40% in idiopathic hypersomnia (IH) and 33-80% in narcolepsy. Per Khan 2015, 69% of NT1, 35% of Narcolepsy Type 2, and 20% of idiopathic hypersomnia (IH). Per Luca 2013, 50-55% in Narcolepsy Type 1.
- Disrupted nighttime sleep — Per ICSD-3-TR for Narcolepsy Type 2: “Nocturnal sleep disruption with frequent awakenings may be present but to a lesser extent than observed in Narcolepsy Type 1.” Per Khan 2015: Narcolepsy Type 1 has significantly lower sleep efficiency than Narcolepsy Type 2 or IH; disrupted sleep may be common in Narcolepsy Type 2 and is not typical in idiopathic hypersomnia (IH).
- Cataplexy — Present by definition in Narcolepsy Type 1 only.
Researchers propose new diagnostic criteria because of these overlaps
In 2020, European researchers published a proposal for new diagnostic criteria for central hypersomnias.
Common misdiagnoses and co-morbidities for idiopathic hypersomnia (IH), Narcolepsy Type 1, and Narcolepsy Type 2
Although the diagnostic criteria for each of these requires that another disorder doesn’t better explain the symptoms, the presence of other disorders doesn’t rule out the hypersomnia diagnosis if EDS continues after adequate treatment of the other disorder. Also, if the hypersomnia predates the other disorder, the other disorder is more likely to be a comorbidity than a replacement for the hypersomnia diagnosis.
Some of the most common misdiagnoses and co-morbidities are with these conditions:
Sleep apnea
Sleep apnea is more common in people with hypersomnias than in the general population and may be the cause of hypersomnolence or comorbid with it. If sleep apnea is present, treat it as well as possible for at least 3 months before diagnosing a comorbid hypersomnia disorder.
Depression
Depression may be the cause of hypersomnolence or comorbid with it, but depression alone shouldn’t cause abnormal MSLT results.
Hypersomnia associated with a mental disorder
Hypersomnolence in people with mental disorders is not necessarily caused by the mental disorder. For example, depression and ADHD are more common in people with hypersomnias than in the general population.
A person diagnosed with hypersomnia associated with depression must have both hypersomnolence and depression, and the two must be related. However, researchers don’t yet know whether the depression causes the hypersomnolence, the hypersomnolence causes the depression, or the two are related in some other way.
Chronic fatigue syndrome (CFS)
People with CFS have persistent or relapsing fatigue that doesn’t go away with sleep or rest. They clearly have fatigue rather than EDS, and they don’t have abnormal MSLTs or other problems with their sleep. CFS can be comorbid with a hypersomnia, but since fatigue can also be a symptom of hypersomnias, this can be hard to differentiate.
Autonomic disorders
About a third of childhood primary hypersomnias are associated with symptoms of significant autonomic dysfunction, such as orthostatic intolerance, palpitations, headache, and dizziness. These can be mistaken as symptoms of the primary sleep disorder.
These symptoms are recognizable at the time of presentation, but they may be masked once hypersomnia medicines are started. Therefore, screen at the initial presentation of the sleep disorder, using autonomic reflex screens and focused questioning. Read more in this 2021 study.
Adults with hypersomnias may also have increased rates of autonomic dysfunction, including POTS (postural orthostatic tachycardia syndrome), that would benefit from early screening.
Hypersomnia associated with a medical disorder
Make this diagnosis instead of idiopathic hypersomnia (IH), Narcolepsy Type 1, or Narcolepsy Type 2 if you find that another medical condition is the primary cause of EDS. Hypersomnolence is associated with many conditions, including:
- Infectious diseases, such as Whipple’s disease and Lyme disease
- Encephalitis and post-infectious syndromes
- Neurodegenerative diseases, such as multiple sclerosis, multiple system atrophy, Parkinson’s, Alzheimer’s, and dementia with lewy bodies
- Neurosarcoidosis
- Brain tumors
- Head trauma
- Stroke
- Epilepsy
- Migraine
- Metabolic encephalopathy, including hepatic, renal, adrenal, pancreatic, toxic, and inherited
- Systemic inflammation, such as from chronic infections, rheumatologic disorders, or cancer
- Autoimmune or paraneoplastic disorders associated with anti-Ma-2 or anti-aquaporin-4 antibodies
- POTS
- Fibromyalgia
- Endocrine disorders, including hypothyroid
- Rare genetic disorders, such as Ehlers-Danlos syndrome, Prader-Willi syndrome, myotonic dystrophy, fragile X, Moebius syndrome, Niemann Pick type C disease, DNMT1 mutations, Williams syndrome, Smith-Magenis syndrome, and Norrie disease
It’s especially important to consider these medical conditions when the hypersomnia diagnosis isn’t clear.
Narcolepsy Type 1 and Narcolepsy Type 2 associated with a medical condition
In these subtypes, although symptoms are due to a medical condition, the full criteria for Narcolepsy Type 1 or Narcolepsy Type 2 are also met. Therefore, the diagnosis is Narcolepsy Type 1 or Narcolepsy Type 2 associated with a medical condition (not hypersomnia associated with a medical condition). This can be very important for access to narcolepsy treatments.
Intermediate (110 to 200 pg/ml) CSF orexin levels have been found in some people with hypersomnolence and encephalopathies or immune-mediated demyelinating disorders. However, orexin levels are not always decreased in Narcolepsy Type 1 due to a medical condition, suggesting different etiologies.
Research in this area continues to evolve and affect classification.
Idiopathic hypersomnia
The cause of idiopathic hypersomnia (IH) isn’t known, and there may be different causes for different groups of people with it. People with idiopathic hypersomnia (IH) have normal CSF orexin levels.
Studies have reported possible causes, including:
- Longer circadian period length, which helps explain the tendency to evening chronotype (being a “night owl”) and difficulties in awakening
- Overproduction of an endogenous peptide in the CSF that acts like a sleeping pill by increasing GABA-A receptor activity
- Disruption of the default-mode network, which was suggested by MRI showing localized differences in brain volume, cortical thickness, and resting functional connectivity (the latter correlated with subjective daytime sleepiness)
- Lower sleep quality, such as from
- Disrupted sleep with numerous arousals
- Less deep (slow wave) sleep
- Autoimmune reaction to infection
- Inflammation
Narcolepsy type 2
The cause of Narcolepsy Type 2 isn’t known, and there may be different causes for different groups of people with it.
- Some people will be reclassified with Narcolepsy Type 1 once they develop cataplexy or low CSF orexin
- Others may not be distinguishable from people with IH because SOREMPs do not seem to be a stable trait in Narcolepsy Type 2 or idiopathic hypersomnia (IH)
- Some would be reclassified with insufficient sleep syndrome when applying strict diagnostic testing protocols
Narcolepsy Type 1
Narcolepsy Type 1 is caused by problems with orexin signaling, most likely due to loss of hypothalamic orexin-producing neurons from an autoimmune process. T cell-mediated destruction of these neurons is most likely. The role of B cells and autoantibodies is unclear, so classical criteria for an autoimmune disorder are not all met.
Animal models show a causal relationship between orexin loss and narcolepsy.
- About 95% of people with narcolepsy and typical cataplexy have undetectable or low CSF orexin levels.
- The other 5% with normal CSF orexin levels suggest that CSF levels do not perfectly reflect orexin neurotransmission or that narcolepsy with cataplexy can have another cause. Many cases without orexin deficiency are familial or secondary to other conditions.
Possible triggers include beta-hemolytic streptococcus and H1N1 influenza. However, neither causal associations nor mechanisms have been firmly established.
- Researchers have found Narcolepsy Type 1 after H1N1 infection or vaccination
- In 2010, researchers reported an increased incidence of Narcolepsy Type 1 following H1N1 vaccination with Pandemrix
- Other research has reported an increase in antibodies against beta-hemolytic strep, which were highest around the onset of narcolepsy
Kleine-Levin syndrome
The cause of KLS isn’t known. However, studies suggest that a localized but multifocal encephalopathy occurs during episodes. The recurrent aspect and the adolescent onset (often occurring with an infection) suggests an autoimmune cause. There may also be circadian, genetic, or metabolic abnormalities.
Brain functional imaging is abnormal in most cases, showing:
- Low perfusion and low metabolism in the thalamus, hypothalamus, posterior associative cortex, and hippocampus
- Occasional high metabolism in the frontal lobe
- Abnormalities happen during episodes and often between episodes as well
Some studies have reported CSF orexin levels one third lower (but still in the normal range) during symptomatic versus asymptomatic periods. CSF cytology and protein are normal.
Risk factors include:
- Birth and developmental problems (found in one third)
- Gene polymorphisms in the TRANK1 region of chromosome 3 (only in the presence of a perinatal injury)
- Ashkenazi descent
Visit our CME page for more information.
American Academy of Sleep Medicine. International Classification of Sleep Disorders, 3rd ed.; Text Revision; American Academy of Sleep Medicine: Darien, IL, USA, 2023.
Barateau, Lucie, et al. “Narcolepsy severity scale-2 and idiopathic hypersomnia severity scale to better quantify symptoms severity and consequences in narcolepsy type 2.” SLEEP, 2024, https://doi.org/10.1093/sleep/zsad323.
Billiard, Michel, and Karel Sonka. “Idiopathic hypersomnia.” Sleep Medicine Reviews, vol. 29, 2016, pp. 23–33, https://doi.org/10.1016/j.smrv.2015.08.007.
Broughton, R., et al. “Ambulatory 24 Hour sleep-wake monitoring in narcolepsy-cataplexy compared to matched controls.” Electroencephalography and Clinical Neurophysiology, vol. 70, no. 6, 1988, pp. 473–481, https://doi.org/10.1016/0013-4694(88)90145-9.
Chervin, Ronald. “Approach to the patient with excessive daytime sleepiness.” UpToDate, 2023.
Chervin, Ronald. “Idiopathic hypersomnia.” UpToDate, 2022.
Cook, J.D., et al. “Identifying subtypes of hypersomnolence disorder: A clustering analysis.” Sleep Medicine, vol. 64, 2019, pp. 71–76, https://doi.org/10.1016/j.sleep.2019.06.015.
Diagnostic and Statistical Manual of Mental Disorders: FITFH Edition, Text Revision: DSM-5-TR. American Psychiatric Association Publishing, 2022.
Fronczek, Rolf, and Gert Jan Lammers. “Narcolepsy type 1: Should we only target hypocretin receptor 2?” Clinical and Translational Neuroscience, vol. 7, no. 3, 2023, p. 28, https://doi.org/10.3390/ctn7030028
Gool, Jari K., et al. “Data-driven phenotyping of central disorders of hypersomnolence with unsupervised clustering.” Neurology, vol. 98, no. 23, 2022, https://doi.org/10.1212/wnl.0000000000200519.
Jagadish, Spoorthi, et al. “Autonomic dysfunction in childhood hypersomnia disorders.” Sleep Medicine, vol. 78, 2021, pp. 43–48, https://doi.org/10.1016/j.sleep.2020.11.040.
Kawai, Makoto, et al. “Narcolepsy in African Americans.” Sleep, vol. 38, no. 11, 2015, pp. 1673–1681, https://doi.org/10.5665/sleep.5140.
Khan, Zeeshan, and Lynn Marie Trotti. “Central Disorders of hypersomnolence.” Chest, vol. 148, no. 1, 2015, pp. 262–273, https://doi.org/10.1378/chest.14-1304.
Krahn, Lois E., et al. “Recommended protocols for the multiple sleep latency test and maintenance of wakefulness test in adults: Guidance from the American Academy of Sleep Medicine.” Journal of Clinical Sleep Medicine, vol. 17, no. 12, 2021, pp. 2489–2498, https://doi.org/10.5664/jcsm.9620.
Lammers, Gert Jan, et al. “Diagnosis of central disorders of hypersomnolence: A reappraisal by European experts.” Sleep Medicine Reviews, vol. 52, 2020, p. 101306, https://doi.org/10.1016/j.smrv.2020.101306.
Lecendreux, Michel, et al. “Attention-deficit/hyperactivity disorder (ADHD) symptoms in pediatric narcolepsy: A cross-sectional study.” Sleep, vol. 38, no. 8, 2015, pp. 1285–1295, https://doi.org/10.5665/sleep.4910.
Luca, Gianina, et al. “Clinical, polysomnographic and genome‐wide association analyses of narcolepsy with cataplexy: A european narcolepsy network study.” Journal of Sleep Research, vol. 22, no. 5, 2013, pp. 482–495, https://doi.org/10.1111/jsr.12044.
Malhotra, Raman K. “Evaluating the sleepy and sleepless patient.” CONTINUUM: Lifelong Learning in Neurology, vol. 26, no. 4, 2020, pp. 871–889, https://doi.org/10.1212/con.0000000000000880.
Maness, Caroline, et al. “Systemic exertion intolerance disease/chronic fatigue syndrome is common in sleep centre patients with hypersomnolence: A retrospective pilot study.” Journal of Sleep Research, vol. 28, no. 3, 2018, https://doi.org/10.1111/jsr.12689.
Maski, Kiran, et al. “Listening to the patient voice in narcolepsy: Diagnostic Delay, disease burden, and treatment efficacy.” Journal of Clinical Sleep Medicine, vol. 13, no. 03, 2017, pp. 419–425, https://doi.org/10.5664/jcsm.6494.
Maski, Kiran. “Understanding racial differences in narcolepsy symptoms may improve diagnosis.” Sleep, vol. 38, no. 11, 2015, pp. 1663–1664, https://doi.org/10.5665/sleep.5130.
Miano, Silvia, et al. “A series of 7 cases of patients with narcolepsy with hypocretin deficiency without the HLA DQB1*06:02 allele.” Journal of Clinical Sleep Medicine, vol. 19, no. 12, 2023, pp. 2053–2057, https://doi.org/10.5664/jcsm.10748.
Miglis, Mitchell G., et al. “Frequency and severity of autonomic symptoms in idiopathic hypersomnia.” Journal of Clinical Sleep Medicine, vol. 16, no. 5, 2020, pp. 749–756, https://doi.org/10.5664/jcsm.8344.
Murray, Brian James. “Excessive daytime sleepiness due to medical disorders and medications.” UpToDate, 2021.
Šonka, Karel, et al. “Narcolepsy with and without cataplexy, idiopathic hypersomnia with and without long sleep time: A cluster analysis.” Sleep Medicine, vol. 16, no. 2, 2015, pp. 225–231, https://doi.org/10.1016/j.sleep.2014.09.016.
Trotti, Lynn Marie. “Central Disorders of hypersomnolence.” CONTINUUM: Lifelong Learning in Neurology, vol. 26, no. 4, 2020, pp. 890–907, https://doi.org/10.1212/con.0000000000000883.
Trotti, Lynn Marie. “Idiopathic hypersomnia.” Sleep Medicine Clinics, vol. 12, no. 3, 2017, pp. 331–344, https://doi.org/10.1016/j.jsmc.2017.03.009.
Vernet, Cyrille, and Isabelle Arnulf. “Narcolepsy with long sleep time: A specific entity?” Sleep, vol. 32, no. 9, 2009, pp. 1229–1235, https://doi.org/10.1093/sleep/32.9.1229.
Zhang, Min, et al. “Narcolepsy with cataplexy: Does age at diagnosis change the clinical picture?” CNS Neuroscience & Therapeutics, vol. 26, no. 10, 2020, pp. 1092–1102, https://doi.org/10.1111/cns.13438.
