SomnusNooze

Why do Emory University’s sleep researchers perform spinal tap procedures on sleepy patients? What is the “sleepy juice” they’re looking for in the spinal fluid? Does it have anything to do with prostaglandins? An expert answers all these questions! We are delighted to have Dr. Amanda Freeman, a PhD neuroscientist and professor at Emory University, who has extensive experience in researching GABA-related hypersomnias, answer these questions about spinal fluid and prostaglandins, and whether these substances might help unlock the mysteries of hypersomnias.
QUESTION:
What exactly are sleep researchers (such as Dr. David Rye and his team at Emory) looking for in the spinal taps they perform on persons with suspected hypersomnias? What is the “sleepy juice” that such researchers talk about? And does it have anything to do with prostaglandins? I have heard that prostaglandins are in spinal fluid and in the GABA pathway (and can sometimes be detected in certain types of urine samples), but do prostaglandins play any part in hypersomnias?
ANSWER:
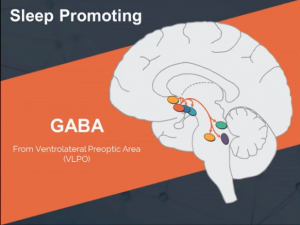
A spinal tap is performed in order to collect cerebrospinal fluid (CSF) which is produced by specialized cells in the brain. CSF surrounds the brain and spinal cord and functions to cushion the central nervous system, as well as to provide nutrients and to remove waste. Due to the close contact with the brain, the composition of the CSF can provide insight on the neural processes. The same information is not available in blood samples, because a physical barrier separates the CSF from the blood and filters out these biomarkers. Dr. Rye and his team are looking at CSF in order to identify the specific substance, present in people with IH, that increases the inhibitory response of GABAA receptors1. As you may recall from the first Learning About GABA video, GABA promotes sleep by inhibiting all of the wake-promoting nuclei; therefore, the substance that increases the inhibitory GABAA receptor-mediated current likely drives the sleepiness experienced by IH patients. CSF is a clear fluid that contains many different substances, including proteins, ions, enzymes, and other compounds. Since the specific IH-related substance has yet to be identified, Dr. Rye began referring to the CSF from IH patients as “sleepy juice,” because somewhere in that fluid is the substance that drives the increased sleep need.
 Whatever this IH-related substance is, it is likely not a prostaglandin. In an effort to characterize the stability of the IH-related substance, Dr. Rye and his team have compared the effects of CSF samples immediately after thawing to those kept at room temperature for 72 hours. The team observed similar magnitudes of increased GABAA receptor-mediated current under both conditions1. Prostaglandins, however, are unstable at room temperature and would not be expected to retain the same level of activity after the room temperature exposure. In addition, Dr. Rye’s team estimates that the size of the IH-related substance is 500- to 3000-daltons1, which is larger than most prostaglandins (300- to 400-daltons)2.
Whatever this IH-related substance is, it is likely not a prostaglandin. In an effort to characterize the stability of the IH-related substance, Dr. Rye and his team have compared the effects of CSF samples immediately after thawing to those kept at room temperature for 72 hours. The team observed similar magnitudes of increased GABAA receptor-mediated current under both conditions1. Prostaglandins, however, are unstable at room temperature and would not be expected to retain the same level of activity after the room temperature exposure. In addition, Dr. Rye’s team estimates that the size of the IH-related substance is 500- to 3000-daltons1, which is larger than most prostaglandins (300- to 400-daltons)2.
It is possible, however, that prostaglandins play a role in other forms of hypersomnia. Prostaglandins are lipid-based substances that are naturally synthesized in the body. They are pro-inflammatory and play a role in generating the immune response. When we think of inflammation, we often recall the response to an injury, such as scraping your knee, and the characteristic inflammatory response of redness, heat, and swelling. An inflammatory response, however, can also include behavioral effects. For example, when someone is sick with the flu and has a fever, there are corresponding changes in behavior, including lack of energy, loss of appetite, decreased activity, and loss of interest in social engagement. These “sickness behaviors” are consequences of the immune response to the infection and are mediated, at least in part, by prostaglandins3. There are several subtypes of prostaglandins, but several lines of evidence indicate that prostaglandin D2 (PGD2) is primarily responsible for driving the increase in sleepiness. For example, when PGD2 is experimentally infused into the brain of rats, it induces sleep4. Naturally-occurring levels of PGD2 in the brain demonstrate a circadian rhythm5, and sleep deprivation drives an increase in PGD2 levels6. These sleep-promoting effects occur when PGD2 binds to the prostaglandin D2 receptor 1 (DP1), which results in increases in adenosine levels and, ultimately, GABA levels7. In fact, recent studies have found that the autoimmune responses targeting the DP1 receptor may contribute to the disrupted sleep/wake regulation in narcolepsy type 18.
Disclaimer for Ask the Doctor: The medical information provided is intended for educational purposes only and is not a substitute for professional medical care or advice. Every person is different, and questions about your own personal health conditions should be discussed with your own healthcare professional.
Works cited
1 Rye, D.B., Bliwise, D.L., Parker, K., Trotti, L.M., Saini, P., Fairley, J., Freeman, A., Garcia, P.S., Owens, M.J., Ritchie, J.C., et al. (2012). Modulation of vigilance in the primary hypersomnias by endogenous enhancement of GABAA receptors. Sci Transl Med 4, 161ra151.
2 Andersson, H. (2008). Chapter 21 – Clinical Reproductive Endocrinology. In Clinical Biochemistry of Domestic Animals (Sixth Edition), J.J. Kaneko, J.W. Harvey, and M.L. Bruss, eds. (San Diego: Academic Press), pp. 635-662.
3 Pecchi, E., Dallaporta, M., Jean, A., Thirion, S., and Troadec, J.D. (2009). Prostaglandins and sickness behavior: old story, new insights. Physiol Behav 97, 279-292.
4 Ueno, R., Ishikawa, Y., Nakayama, T., and Hayaishi, O. (1982). Prostaglandin D2 induces sleep when microinjected into the preoptic area of conscious rats. Biochem Biophys Res Commun 109, 576-582.
5 Pandey, H.P., Ram, A., Matsumura, H., and Hayaishi, O. (1995). Concentration of prostaglandin D2 in cerebrospinal fluid exhibits a circadian alteration in conscious rats. Biochem Mol Biol Int 37, 431-437.
6 Ram, A., Pandey, H.P., Matsumura, H., Kasahara-Orita, K., Nakajima, T., Takahata, R., Satoh, S., Terao, A., and Hayaishi, O. (1997). CSF levels of prostaglandins, especially the level of prostaglandin D2, are correlated with increasing propensity towards sleep in rats. Brain Res 751, 81-89.
7 Hayaishi, O. (2000). Molecular mechanisms of sleep-wake regulation: a role of prostaglandin D2. Philos Trans R Soc Lond B Biol Sci 355, 275-280.
8 Sadam, H., Pihlak, A., Kivil, A., Pihelgas, S., Jaago, M., Adler, P., Vilo, J., Vapalahti, O., Neuman, T., Lindholm, D., et al. (2018). Prostaglandin D2 Receptor DP1 Antibodies Predict Vaccine-induced and Spontaneous Narcolepsy Type 1: Large-scale Study of Antibody Profiling. EBioMedicine 29, 47-59.
Additional References
Barmashenko, G., Schmidt, M., and Hoffmann, K.P. (2005). Differences between cation-chloride co-transporter functions in the visual cortex of pigmented and albino rats. Eur J Neurosci 21, 1189-1195.
Blaszczyk, W.M., Straub, H., and Distler, C. (2004). GABA content in the retina of pigmented and albino rats. Neuroreport 15, 1141-1144.
Burbridge, T., Choudhury, B.P., and Collard, K.J. (1996). The uptake of gamma-aminobutyric acid and glutamate by synaptosomes from the visual cortex of albino and pigmented rabbits. J Neural Transm (Vienna) 103, 299-305.
Diykov, D., Barmashenko, G., and Hoffmann, K.P. (2008a). Development of chloride homeostasis in albino and pigmented rat visual cortex neurons. Neuroreport 19, 595-598.
Diykov, D., Turchinovich, A., Zoidl, G., and Hoffmann, K.P. (2008b). Elevated intracellular chloride level in albino visual cortex neurons is mediated by Na-K-Cl co-transporter. BMC Neurosci 9, 57.
Krause, M., and Hoffmann, K.P. (2009). Shift of chloride reversal potential in neurons of the accessory optic system in albinotic rats. Exp Brain Res 199, 345-353.
