Medicines for idiopathic hypersomnia and narcolepsy
and avoiding interactions with hormone medicines, such as birth control, hormone replacement therapy, and gender-affirming therapy

and avoiding interactions with hormone medicines, such as birth control, hormone replacement therapy, and gender-affirming therapy
Jump to page sections
The table below shows medicines that are approved by the U.S. Federal Drug Administration (FDA) specifically to treat narcolepsy (*) or IH (†) and symptoms the medicines may improve. In the U.S., as long as a medicine is approved by the FDA to treat any condition, doctors may prescribe it “off-label” to treat other conditions.
| Generic medicine names (example brand names) | Daytime sleepiness | Cataplexy | Sleep inertia | Sleep disruption |
|---|---|---|---|---|
| Modafinils* (such as Nuvigil) | ||||
| Oxybates*† (such as Xyrem) | ||||
| Pitolisant* (such as Wakix) | ||||
| Solriamfetol* (such as Sunosi) | ||||
| Amphetamines* (such as Adderall) | ||||
| Methylphenidates* (such as Jornay PM) |
The table below shows other medicines that may be used to treat IH or narcolepsy, and the symptoms they may improve, based on currently available evidence. None of these are FDA-approved specifically to treat IH or narcolepsy.
| Generic medicine names (example brand names) | Daytime sleepiness | Cataplexy | Sleep inertia | Sleep disruption |
|---|---|---|---|---|
| Clarithromycin (such as Biaxin) | ||||
| Flumazenil | ||||
| Antidepressants (such as Prozac) | ||||
| Bupropion (such as Wellbutrin) | ||||
| Bupropion long-acting (such as Wellbutrin XL) | ||||
| Protriptyline (such as Vivactil) | ||||
| Selegiline (such as Emsam) | ||||
| Atomoxetine (such as Strattera) | ||||
| Reboxetine (such as Davedex) | ||||
| Levothyroxine (such as Synthroid) | ||||
| Baclofen (such as Lioresal) | ||||
| Caffeine (such as No Doz, drinks) | ||||
| Carnitine supplements (such as Carnitor) | ||||
| Melatonin long-acting |
For more in-depth information about these medicines, such as side effects, mechanisms of action, and evidence for use, visit our healthcare professional web page, “Treating people who have idiopathic hypersomnia or narcolepsy.”
Currently, there are no medicines that specifically treat brain fog or automatic behaviors. However, treating daytime sleepiness may also help these symptoms.
Visit Project Sleep’s “Sleep Paralysis and Hallucinations” web page to get some other ideas and understanding that can help with these symptom
 You may need to try different hypersomnia medicines and doses to find the treatment that works best for you
You may need to try different hypersomnia medicines and doses to find the treatment that works best for youAs you work with your doctor to try different hypersomnia medicines, here are some things to think about.
Some medicines work best when you take them at a specific time of day. Ask your doctor how many hours each medicine usually takes to start working and how long they’ll last during the day. This can help you plan when to take them.
Some medicines may have side effects, such as nausea, headaches, or raised blood pressure. Talk with your doctor about which side effects are the most common and which are the most dangerous. Ask about any side effects that worry you.
You may have no side effects at all, but there’s usually no way to find out without trying the medicine yourself.
![]() If you could become pregnant, talk with your doctor about the safety of your medicine during pregnancy and nursing. Visit our web page “Pregnancy and safe baby care.”
If you could become pregnant, talk with your doctor about the safety of your medicine during pregnancy and nursing. Visit our web page “Pregnancy and safe baby care.”
Learn more about side effects:
Many of the more common or important side effects are listed on our Treatment web page for doctors, which includes more in-depth information about hypersomnia medicines
Talk with your doctor about starting with a low dose and slowly raising it. This may help you to get a feel for:
You may need different doses for different days, such as:
If you menstruate (have a period):
If you’re taking a combination of medicines and need to make changes, it’s usually best to change just one medicine at a time. Talk with your doctor before making any changes.
You still may not feel fully alert, even if you’re taking the medicines that work best for you. A good goal is to try to be most alert at important times of the day, such as during work, school, or while driving.
The Idiopathic Hypersomnia Severity Scale (IHSS) and Narcolepsy Severity Scale (NSS) are tools that you and your doctors can use to help measure how well a treatment is working to ease your symptoms. Depending on your diagnosis, you can take one of these questionnaires before and after you start a new treatment to see if your scores improve. The FOSQ (Functional Outcomes of Sleep Questionnaire) and ESS (Epworth Sleepiness Scale) are other options for people with any hypersomnia sleep disorder.
Getting support from other people with hypersomnias can help. You can join support groups (visit the Support group section on our “Quality of life tips” web page) or ask your doctor if they can give your contact information to other people who have taken the same medicines as you.
These hypersomnia medicines can affect how well medicines with hormones (estrogens, progestins, or testosterones) work:
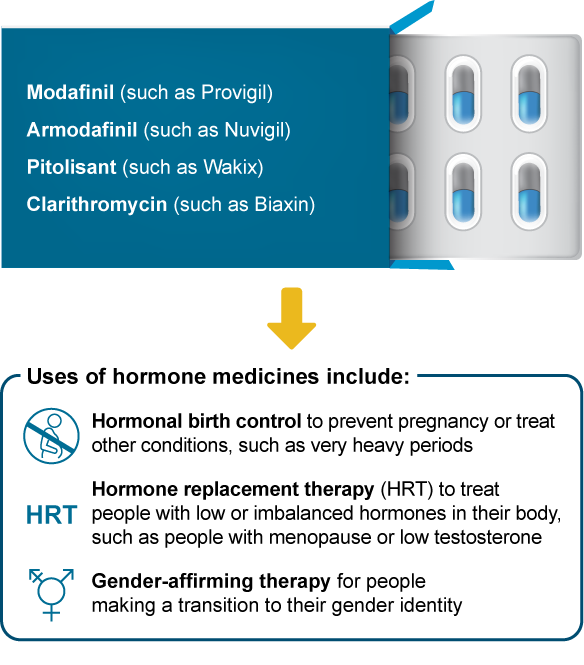
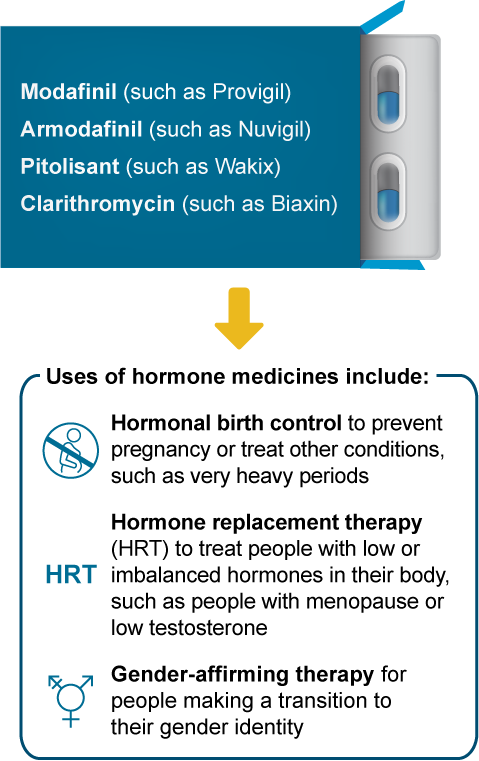
What may happen if I take these medicines together?
Less symptom control, such as not controlling hot flashes during menopause
A higher risk of pregnancy
Less medicine effectiveness, such as with gender-affirming therapy
More side effects, such as headaches or breakthrough menstrual bleeding
It’s usually safe to use these hypersomnia medicines with hormone medicines. To help ensure the hormone medicine still works well, you and your doctor may decide to:

|
Hormonal birth control options that can interact with hypersomnia medicines include: |
| Birth control implant |
| Birth control pills |
| Birth control patch |
| Birth control ring |
| Emergency birth control |
|
Other birth control options that don’t interact with hypersomnia medicines include: |
| Sterilization (vasectomy or tubal ligation) |
| IUDs |
| Progestin shots (such as Depo-Provera) |
| Condoms |
| Contraceptive diaphragms |
For more in-depth information on managing interactions between hypersomnia and hormone medicines, see the section “Some hypersomnia medicines interact with hormone medicines,” on our Treatment web page for doctors.
 Volunteering for clinical trials may allow you to try medicines that aren’t yet available in your home country nor approved for your diagnosis. Some clinical trials allow participants to continue taking the medicine for a period of time after the trial has ended. Clinical trials also help scientists better understand hypersomnias and their treatment. For more information:
Volunteering for clinical trials may allow you to try medicines that aren’t yet available in your home country nor approved for your diagnosis. Some clinical trials allow participants to continue taking the medicine for a period of time after the trial has ended. Clinical trials also help scientists better understand hypersomnias and their treatment. For more information:
Some people with hypersomnias may have other health problems that make their symptoms worse. Ask your doctor to screen you for these, such as:
Published Apr. 22, 2020 |
Revised Feb. 1, 2024
Complete update Jan. 19, 2024 |
Approved by our medical advisory board